KPV 10MG
$89.99
Shipping Calculated at checkout.
Availability: 17 in stock
FREE SHIPPING
99%+ PURITY
MADE IN USA
Vial
- Mazdutide 10MG
- AICAR 50MG
- Lipo-C 10ML
- Survodutide 10MG
- LC-526 Metabolic Complex
- SSJ-9 Amino Matrix
- Epitalon 10MG
- L-Carnitine 600MG / 10ML
- Cerebrolysin 60MG
- Oxytocin 5MG
- SELANK
- 5-Amino-1MQ
- VIP
- L-Glutathione 1500MG
- Acetic Acid Solution
- HCG 10000iu
- BAC Water 30ML
- BAC Water 10ML
- GLOW 70MG
- IGF-1 LR3 1MG
- Thymalin
- HCG (5000iu)
- Wolverine Stack Peptides – BPC-157 (5mg) / TB-500 (5mg)
- BAC Water 3ML
- Melanotan II (MT-2) 10MG
- SLU-PP-332 (5MG)
- KPV 10MG
- GHK-Cu
- KLOW 80 – GHK-Cu (50mg) / KPV (10mg) / BPC-157 (10mg) / TB500 (10mg)
- Gonadorelin
- L-Glutathione (200MG)
- SEMAX (10MG)
- Cagrilintide (10MG)
- DSIP – Delta Sleep‑Inducing Peptide (5MG)
- RETA GLP-3
- Kisspeptin-10
- Ipamorelin / CJC-1295 No Dac 10mg
- Tirz GLP-2
- L-Glutathione (500MG)
- Pancragen 20 MG
- Prostamax 20 MG
- Livagen 20 MG
- Testagen 20 MG
- CardioCytogen 20 MG
- TB-4
- ARA‑290 10mg
- FOXO4-DRI (D-Retro-Inverso) 10mg
- SS-31
- Tesamorelin (10MG)
- AOD-9604
- IPAMORELIN 10MG
- PT-141 10MG
- NAD+
- TB-500 10MG
- SEMA GLP-1
- MOTS-c 10MG
- CJC-1295 (10MG)
- BPC-157 (10MG)
Nasal Spray
- Glutathione Spray
- TB-500 Spray
- MT-2 Spray
- MT-1 Nasal Spray
- AOD-9604 Spray
- NAD+ 500MG Spray
- BPC-157 Spray
- DSIP 5MG Spray
- VIP 10MG Spray
- Oxytocin 10MG Spray
- PT-141 10MG Spray
- GHK-Cu Spray
- P21 Peptide Spray
- N-Acetyl Selank Spray
- N-acetyl Semax Spray
- Adamax Nootropic – Nasal Spray
- Adalank Nootropic Peptide – Nasal Spray
IRON
- Dihexa
- Glutathione Spray
- TB-500 Spray
- Reduce (BAM, SLUPP322)
- MT-2 Spray
- MT-1 Nasal Spray
- AOD-9604 Spray
- Phenibut (60 capsules)
- NAD+ 500MG Spray
- BPC-157 Spray
- Mazdutide 10MG
- AICAR 50MG
- Lipo-C 10ML
- Survodutide 10MG
- Methylene Blue 20MG
- 5-Amino-1MQ Capsules 50mg 60 cap
- LC-526 Metabolic Complex
- SSJ-9 Amino Matrix
- MITOCHONDRIAL POWER STACK
- SHRED MATRIX X4
- ANABOLIC SIGNALING MATRIX X3
- L-Carnitine 600MG / 10ML
- Cerebrolysin 60MG
- Oxytocin 5MG
- SELANK
- 5-Amino-1MQ
- VIP
- L-Glutathione 1500MG
- Acetic Acid Solution
- HCG 10000iu
- BAC Water 30ML
- BAC Water 10ML
- GLOW 70MG
- IGF-1 LR3 1MG
- Thymalin
- HCG (5000iu)
- Wolverine Stack Peptides – BPC-157 (5mg) / TB-500 (5mg)
- BAC Water 3ML
- Melanotan II (MT-2) 10MG
- SLU-PP-332 (5MG)
- KPV 10MG
- GHK-Cu
- KLOW 80 – GHK-Cu (50mg) / KPV (10mg) / BPC-157 (10mg) / TB500 (10mg)
- Gonadorelin
- L-Glutathione (200MG)
- SEMAX (10MG)
- Cagrilintide (10MG)
- DSIP – Delta Sleep‑Inducing Peptide (5MG)
- RETA GLP-3
- MK-ULTRA (MK-777)
- Kisspeptin-10
- VIP 10MG Spray
- Ipamorelin / CJC-1295 No Dac 10mg
- Tirz GLP-2
- L-Glutathione (500MG)
- GHK-Cu Spray
- P21 Peptide Spray
- N-Acetyl Selank Spray
- N-acetyl Semax Spray
- Adamax Nootropic – Nasal Spray
- Adalank Nootropic Peptide – Nasal Spray
- Pancragen 20 MG
- Prostamax 20 MG
- Livagen 20 MG
- Testagen 20 MG
- CardioCytogen 20 MG
- TB-4
- ARA‑290 10mg
- FOXO4-DRI (D-Retro-Inverso) 10mg
- KPV 250 MCG (60 Capsules)
- SLU-PP-332 (60 capsules, 250MCG)
- SS-31
- Tesamorelin (10MG)
- MK-677 12.5MG (60 capsules)
- AOD-9604
- IPAMORELIN 10MG
- Healing and Repair Research Blend (60 capsules)
- BPC-157 250MCG (60 capsules)
- Tesofensine 500MCG (100 capsules)
- PT-141 10MG
- NAD+
- TB-500 10MG
- SEMA GLP-1
- MOTS-c 10MG
- CJC-1295 (10MG)
- BPC-157 (10MG)
Capsules
- Dihexa
- Reduce (BAM, SLUPP322)
- Phenibut (60 capsules)
- Methylene Blue 20MG
- 5-Amino-1MQ Capsules 50mg 60 cap
- MK-ULTRA (MK-777)
- KPV 250 MCG (60 Capsules)
- SLU-PP-332 (60 capsules, 250MCG)
- MK-677 12.5MG (60 capsules)
- Healing and Repair Research Blend (60 capsules)
- BPC-157 250MCG (60 capsules)
- Tesofensine 500MCG (100 capsules)

What is KPV 10MG?
Introduction
KPV (Ac-KPV-NH₂) 5 mg is a synthetic research peptide representing the tripeptide fragment Lys-Pro-Val derived from the C-terminal end of alpha-melanocyte-stimulating hormone (α-MSH), designed to retain the anti-inflammatory properties of the parent hormone in a minimal structure. This small peptide is widely used in preclinical and mechanistic studies because it influences key intracellular signaling pathways involved in immune modulation and inflammation without the broader hormonal effects associated with full-length α-MSH. In vitro and in vivo research has demonstrated that KPV can inhibit activation of nuclear factor-κB (NF-κB) and MAP kinase pathways, which are central regulators of pro-inflammatory gene expression, leading to reduced secretion of cytokines such as TNF-α and IL-1β in experimental models of inflammation. (1) In experimental models of intestinal inflammation, KPV shows notable regulatory effects on mucosal integrity and immune responses. For example, studies using chemically induced colitis in mice reported that KPV treatment reduced histological inflammation, decreased myeloperoxidase activity (a marker of neutrophil infiltration), and improved recovery, suggesting potential utility as a molecular tool for investigating barrier protection and inflammatory bowel disease mechanisms. (2) However, it is important to emphasize that KPV remains a research-grade compound and is not approved for clinical use in humans; its applications are limited to controlled laboratory or preclinical settings focused on understanding peptide-mediated regulation of inflammation and epithelial homeostasis.(3)
Chemical Structure of KPV 10MG
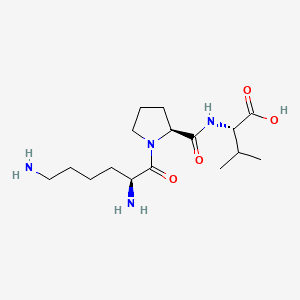
Source; pubchem
Amino Acid Sequence: Lys-Pro-Val
Molecular Formula: C₁₇H₃₂N₆O₄
Molecular Weight: 384.48 g/mol
PubChem CID: 125672
CAS Number: 112965-21-6
Synonyms: MSH (11-13), ACTH(11-13), alpha-MSH(11-13)

What Are the Effects of KPV 10MG?
KPV (Ac-KPV-NH₂) is a synthetic tripeptide retains the potent anti-inflammatory and immunomodulatory properties of the parent hormone while being small, stable, and easily used in experimental studies. KPV is particularly valuable for investigating inflammation, epithelial barrier integrity, and immune cell regulation because it acts at multiple levels of the inflammatory cascade (1,2).
Receptor-Mediated Anti-Inflammatory Signaling
KPV primarily exerts its effects by binding to melanocortin receptors (MC-Rs), especially MC1R, which are expressed on immune cells and epithelial cells. This receptor engagement activates the cyclic AMP (cAMP) signaling pathway, leading to phosphorylation of downstream targets that inhibit the nuclear translocation of NF-κB, a key transcription factor that drives the expression of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6. By dampening NF-κB activity KPV suppresses inflammatory gene transcription and reduces the amplification of immune responses (4).
Intracellular, Receptor-Independent Modulation of NF-κB Pathway
In addition to receptor-mediated effects, KPV can act directly inside cells. Epithelial and immune cells uptake KPV through transporters such as PepT1, allowing it to interact with intracellular signaling proteins. KPV stabilizes IκBα, the inhibitory protein that binds NF-κB in the cytoplasm, preventing NF-κB from entering the nucleus and initiating transcription of inflammatory genes. This dual mechanism both receptor-dependent and intracellular amplifies the peptide’s anti-inflammatory potency (2).
Downstream Biological Effects and Tissue Protection
The combined receptor-mediated and intracellular actions of KPV lead to a range of biological outcomes. These include suppression of pro-inflammatory cytokine release, decreased recruitment of immune cells to inflamed tissues, and enhanced maintenance of epithelial barrier integrity. In experimental models, such as chemically induced colitis or skin inflammation, KPV treatment results in reduced tissue damage, improved epithelial healing, and overall maintenance of tissue homeostasis. These effects make KPV a powerful tool for studying mechanisms of inflammation and for exploring potential therapeutic strategies in preclinical settings (1,2,4).
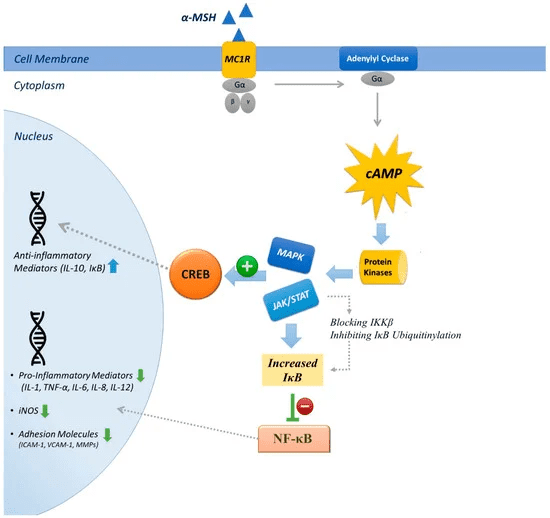
Source; MDPI journal
Research implications
Experimental Intestinal Models for Cytokine and Inflammatory Marker Quantification
Experimental intestinal inflammation models are employed to quantify cytokine-associated signaling and biochemical inflammatory markers following exposure to KPV (Ac-KPV-NH₂). In DSS and TNBS-induced colitis systems, the peptide is examined for its influence on soluble mediators that reflect inflammatory activity within intestinal tissue. Quantitative approaches such as ELISA, multiplex cytokine assays, and real-time PCR are used to measure shifts in concentrations of tumor necrosis factor-α, interleukin-1β, and interleukin-6, alongside downstream inflammatory enzymes including cyclooxygenase-2 and inducible nitric oxide synthase. These measurements allow controlled evaluation of how peptide administration alters cytokine-linked signaling intensity and biochemical inflammatory burden in experimental settings (5,6).
Parallel assessments focus on tissue-level inflammatory indices that accompany cytokine signaling changes. Investigators analyze colon homogenates and histological samples to determine levels of myeloperoxidase activity, nitric oxide derivatives, prostaglandin production, and other inflammatory metabolites that reflect the magnitude of experimental colitis. Correlating these parameters with cytokine quantification enables integrated mapping of inflammatory marker profiles following KPV exposure in laboratory models designed to simulate intestinal inflammatory conditions (7).
PepT1-Mediated Uptake Expression Studies
Epithelial transport studies are used to examine how KPV (Ac-KPV-NH₂) is absorbed through intestinal peptide transport systems and how inflammatory conditions influence this process. Experimental work demonstrates that the proton-coupled oligopeptide transporter PepT1 facilitates cellular entry of the tripeptide across intestinal epithelial membranes, allowing researchers to quantify uptake kinetics, transporter affinity, and intracellular peptide accumulation under controlled laboratory conditions. These investigations typically employ cultured intestinal cell lines and inflamed tissue models to determine how transporter activity changes in response to inflammatory stimuli and how this alters peptide availability within the mucosal environment (5,8).
Such models also enable measurement of inflammation-linked variations in transporter expression and function. Molecular assays, including mRNA expression profiling and protein quantification, are used to assess PepT1 regulation during inflammatory states and to correlate transporter levels with peptide transport efficiency and local biochemical inflammatory indicators. By linking uptake dynamics with transporter expression patterns, these studies provide insight into how KPV distribution within intestinal tissue is shaped by inflammation associated alterations in epithelial transport mechanisms (9).
KPV Modulation of NF‑κB and MAPK Signaling
Cell‑based pathway assays that measure NF‑κB and MAPK activity under inflammatory stimulation are commonly used to evaluate how compounds like KPV modulate key intracellular signaling events triggered by pro‑inflammatory stimuli (e.g., IL‑1β, TNF‑α, LPS). In these assays, cultured human epithelial or immune cells are stimulated with an inflammatory challenge, and pathway reporter systems (like NF‑κB luciferase), protein phosphorylation status, and downstream cytokine output are quantified to assess pathway activation. This functional approach captures multiple signaling layers from receptor engagement through kinase cascades to gene transcription, allowing researchers to determine how KPV affects innate signaling hubs that drive inflammatory gene expression.(10–12)
Intestinal inflammataion
Previous literature demonstrates that KPV significantly attenuates NF‑κB and MAPK activation in vitro and in vivo. In human intestinal epithelial cell models (e.g., Caco2‑BBE), KPV co‑treatment reduced IL‑1β–induced NF‑κB‑dependent luciferase activity, preserved IκB‑α (thereby blocking NF‑κB nuclear translocation), and decreased phosphorylation of MAPK family members such as ERK1/2, JNK, and p38, leading to lowered IL‑8 mRNA and protein levels compared with inflammatory controls.(13) In mouse models of colitis (DSS‑ and TNBS‑induced), oral KPV reduced clinical and histologic inflammation and decreased pro‑inflammatory cytokine expression, indicating that inhibition of NF‑κB/MAPK correlates with reduced disease severity(14).
Another study using human bronchial epithelial cells found KPV suppressed TNF‑α–induced NF‑κB activity and related pro‑inflammatory mediators, supporting broader application beyond gut models.(15) These data illustrate that KPV modulates core inflammatory signaling pathways in cell‑based assays under stimulated conditions, providing mechanistic support for its anti‑inflammatory potential in preclinical research.
Inflammataion associated models
In additional preclinical inflammatory paradigms such as experimental contact dermatitis, researchers have compared the anti‑inflammatory effects of full‑length alpha‑melanocyte‑stimulating hormone (α‑MSH) versus its truncated fragment Ac‑KPV‑NH₂ (the C‑terminal Lys‑Pro‑Val tripeptide). These studies typically measure indicators such as ear swelling, leukocyte infiltration, and cytokine profiles to quantify inflammatory responses following exposure to a sensitizing irritant (16). Both peptides have shown significant suppression of dermatitis‑associated swelling at early timepoints (24 hours), indicating that the shorter Ac‑KPV‑NH₂ peptide can approximate the anti‑inflammatory efficacy of the full hormone immediately after irritant challenge (17). However, in longer term assessments (e.g., at two weeks), the sustained response differs, animals exposed to irritant without continued peptide intervention reveal that α‑MSH maintains a more prolonged reduction in swelling relative to Ac‑KPV‑NH₂, suggesting differences in the duration of anti‑inflammatory action and possibly in mechanisms related to receptor engagement or downstream signaling signatures between the full hormone and its fragment (18). These comparative findings align with broader literature showing that α‑MSH and related tripeptides modulate inflammatory cell activity and cytokine production in models of contact dermatitis and other immune activation settings (16–18).
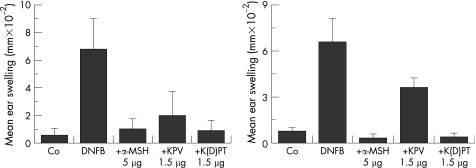
Source; pubmed central
The graph shows that both α‑MSH (5 µg) and KPV (1.5 µg) significantly reduce ear swelling induced by DNFB at 24 hours and 2 weeks compared to DNFB alone, with α‑MSH exhibiting slightly greater suppression at the 2‑week timepoint. Co-treatment with the control peptide K(D)PT has minimal effect, confirming the specificity of the anti-inflammatory response.
Matrix Remodeling Models: Collagen and Fibroblast Signaling
Matrix remodeling models provide controlled systems to study how fibroblasts interact with the extracellular matrix (ECM) particularly collagen under physiological and pathological conditions. These models, including fibroblast-populated collagen lattices and 3D hydrogel systems, allow quantification of collagen deposition, structural organization, and fibroblast-mediated contraction, closely mimicking in vivo tissue remodeling [21,22].
Collagen-Related Endpoints
- Collagen Synthesis and Deposition:
Fibroblasts in these models synthesize type I and III collagen, essential for ECM integrity. Increased collagen deposition, measurable via immunostaining, qPCR, and hydroxyproline assays, reflects fibroblast activation and tissue repair potential. Cytokines such as TGF-β enhance collagen gene expression and matrix deposition, demonstrating reparative remodeling [23,24]. - Matrix Contraction and Organization:
Fibroblast contractility within collagen gels leads to matrix compaction, fiber realignment, and enhanced tensile strength, which can be measured by gel size reduction and imaging of fiber orientation. These endpoints model wound contraction and scar formation (22,25).
- Matrix Degradation:
Matrix metalloproteinases (MMPs), including MMP-2, MMP-9, and MMP-13, regulate collagen turnover. KPV influences fibroblast behavior by modulating MMP activity, balancing ECM degradation and deposition, critical for effective remodeling (26).
Cytokine-Regulated Fibroblast Signaling
Cytokines and growth factors modulate fibroblast behavior in matrix remodeling systems:
- TGF-β induces myofibroblast differentiation, characterized by α-smooth muscle actin (α-SMA) upregulation and increased collagen secretion, enhancing contractile matrix remodeling (23,24).
- PDGF stimulates fibroblast migration and matrix compaction via PI3K and Rho signaling (27).
- Endothelin-1 increases collagen gene expression and suppresses MMP activity, promoting a fibrogenic phenotype (21).
Fibroblast signaling markers, such as phosphorylated kinases and Rho GTPases, can be quantified to assess cellular responses to cytokines and KPV treatment.
Beneficial Effects Observed in Literature
Matrix remodeling studies incorporating KPV have demonstrated several positive effects:
- Enhanced Wound Healing: KPV-treated fibroblasts show increased collagen deposition and contraction, supporting tissue repair (22,23).
- Balanced ECM Remodeling: Modulation of MMP activity by KPV promotes effective ECM turnover without excessive degradation (26).
- Mechanistic Insights: Cytokine-modulated fibroblast signaling elucidates pathways controlling ECM synthesis and contraction, aiding therapeutic development (27).
- Aging and Disease Models: KPV maintains fibroblast function and ECM integrity in aged or stressed systems, reducing pathological fibrosis (25)
References
- Dalmasso G, Charrier-Hisamuddin L, et al. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. Year;Volume(Issue):Page-Page. PMID:18061177.
- Kannengiesser K, Maaser C, Heidemann J, et al. Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease. Inflamm Bowel Dis. 2008 Mar;14(3):324-331.
- KPV tripeptide (Lys-Pro-Val) profile and research overview. PepCodex. Available from: peptcodex.com/peptides/kpv.
- Getting SJ. Targeting melanocortin receptors as a novel strategy to control inflammation. Tendencies Pharmacol Sci. 2006;27(7):316-323.\
- Maaser C, Kannengiesser K, Specht C, et al. Crucial role of the peptide transporter PepT1 for uptake of the anti-inflammatory peptide KPV in intestinal epithelial cells. Gastroenterology. 2006;130(6):1669-1679.
- Brzoska T, Luger TA, Maaser C, Abels C, Böhm M. α-MSH and related tripeptide KPV in experimental inflammatory models. Ann N Y Acad Sci. 2003;994:378-387.
- Getting SJ. Melanocortin peptides and their receptors as modulators of inflammatory processes. Trends Pharmacol Sci. 2002;23(9):447-449.
- Adibi SA. Regulation of expression of the intestinal oligopeptide transporter (PepT1) in health and disease. Am J Physiol Gastrointest Liver Physiol. 2003;285(5):G779-G788.
- Brandsch M. Transport of drugs by proton-coupled peptide transporters: pearls and pitfalls. Expert Opin Drug Metab Toxicol. 2009;5(8):887-905.
- Hayden MS, Ghosh S. NF-κB in immunobiology. Cell Res. 2011;21(2):223–44.
- Cargnello M, Roux PP. Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev. 2011;75(1):50–83.
- Nelson DE, Ihekwaba AE, Elliott M, Gibney CA, Foreman BE, Nelson G, et al. Oscillations in NF-κB signaling control the dynamics of gene expression. 2004;306(5696):704–8.
- Dalmasso G, et al. PepT1‑mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008;134(1):166–78.
- Kannengiesser K, et al. Melanocortin‑derived tripeptide KPV has anti‑inflammatory potential in murine models of inflammatory bowel disease. Inflamm Bowel Dis. 2008;14(3):324–31.
- Inhibition of cellular and systemic inflammation cues … (HBE cells) shows suppressed NF‑κB signaling by KPV.
- Hiltz ME, Lipton JM. Alpha‑MSH peptides inhibit acute inflammation and contact sensitivity. 1990;11(5):979–82. (pubmed.ncbi.nlm.nih.gov)
- Luger TA, Brzoska T. α‑MSH related peptides: a new class of anti‑inflammatory and immunomodulating drugs. Ann Rheum Dis. 2007;66 Suppl 3:iii52–5. (ncbi.nlm.nih.gov)
- Getting SJ, et al. Terminal signal: anti‑inflammatory effects of α‑melanocyte‑stimulating hormone related peptides beyond the pharmacophore. J Endocrinol. 2011;209(3):249–59. (ncbi.nlm.nih.gov)
- https://pmc.ncbi.nlm.nih.gov/articles/PMC2095288/#!po=3.33333
- https://www.mdpi.com/1424-8247/14/1/45
- Shi-Wen X, et al. Fibroblast matrix gene expression: role of endothelin-1. J Invest Dermatol. 2001;117(4):951‑959.
- Rhee S, Grinnell F. Fibroblast mechanics in 3D collagen matrices. Adv Drug Deliv Rev. 2007;59(13):1293‑1300.
- Hinz B, et al. Myofibroblast formation and function in collagen matrices. Nat Rev Mol Cell Biol. 2007;8:657‑667.
- Chen K, et al. MMPs in ECM remodeling in regenerative medicine. Tissue Eng Regen Med. 2023;20:125‑138.
- Nair KG, et al. Tumor secretome drives collagen remodeling via fibroblasts. iScience. 2022;25(8):104637.
- Yu Q, Stamenkovic I. Cell surface-localized MMP-9 regulates collagen remodeling. J Biol Chem. 2000;275(24):18446‑18452.
- Sabeh F, et al. Protease-dependent versus independent cancer cell invasion programs. Nat Cell Biol. 2004;6:712‑722.
https://onlinelibrary.wiley.com/doi/full/10.1111/exd.13887
 KPV 10MG
KPV 10MG
| 5 star | 0% | |
| 4 star | 0% | |
| 3 star | 0% | |
| 2 star | 0% | |
| 1 star | 0% |
Sorry, no reviews match your current selections

TrustScore 4.3

Edgar Guzman
2026-01-20
I have been taking glp3 for a few…
I have been taking glp3 for a few months and I've been losing about 38 close to 40 lb. I feel great. Lots of energy. I definitely recommend iron. It's to me the most trusted legit company customer service It's amazing. They always have discounts. They always have promotions so if you want to see results go with IRON that's for sure
JC
Jcoop
2026-01-16
Amazing service top-notch products
Amazing service top-notch products, I always receive my peptides within a few days of placing my order.
PV
Paola Vargas
2026-01-16
I have try peptides Reta and it’s so…
I have try peptides Reta and it’s so good helping me with better habits and eating well I feel more energy and my progress it’s easier , very focus and the guiadance on usage it’s so good any questions they are there for you . Def recommend
Vial
- Mazdutide 10MG
- AICAR 50MG
- Lipo-C 10ML
- Survodutide 10MG
- LC-526 Metabolic Complex
- SSJ-9 Amino Matrix
- Epitalon 10MG
- L-Carnitine 600MG / 10ML
- Cerebrolysin 60MG
- Oxytocin 5MG
- SELANK
- 5-Amino-1MQ
- VIP
- L-Glutathione 1500MG
- Acetic Acid Solution
- HCG 10000iu
- BAC Water 30ML
- BAC Water 10ML
- GLOW 70MG
- IGF-1 LR3 1MG
- Thymalin
- HCG (5000iu)
- Wolverine Stack Peptides – BPC-157 (5mg) / TB-500 (5mg)
- BAC Water 3ML
- Melanotan II (MT-2) 10MG
- SLU-PP-332 (5MG)
- KPV 10MG
- GHK-Cu
- KLOW 80 – GHK-Cu (50mg) / KPV (10mg) / BPC-157 (10mg) / TB500 (10mg)
- Gonadorelin
- L-Glutathione (200MG)
- SEMAX (10MG)
- Cagrilintide (10MG)
- DSIP – Delta Sleep‑Inducing Peptide (5MG)
- RETA GLP-3
- Kisspeptin-10
- Ipamorelin / CJC-1295 No Dac 10mg
- Tirz GLP-2
- L-Glutathione (500MG)
- Pancragen 20 MG
- Prostamax 20 MG
- Livagen 20 MG
- Testagen 20 MG
- CardioCytogen 20 MG
- TB-4
- ARA‑290 10mg
- FOXO4-DRI (D-Retro-Inverso) 10mg
- SS-31
- Tesamorelin (10MG)
- AOD-9604
- IPAMORELIN 10MG
- PT-141 10MG
- NAD+
- TB-500 10MG
- SEMA GLP-1
- MOTS-c 10MG
- CJC-1295 (10MG)
- BPC-157 (10MG)
Nasal Spray
- Glutathione Spray
- TB-500 Spray
- MT-2 Spray
- MT-1 Nasal Spray
- AOD-9604 Spray
- NAD+ 500MG Spray
- BPC-157 Spray
- DSIP 5MG Spray
- VIP 10MG Spray
- Oxytocin 10MG Spray
- PT-141 10MG Spray
- GHK-Cu Spray
- P21 Peptide Spray
- N-Acetyl Selank Spray
- N-acetyl Semax Spray
- Adamax Nootropic – Nasal Spray
- Adalank Nootropic Peptide – Nasal Spray
IRON
- Dihexa
- Glutathione Spray
- TB-500 Spray
- Reduce (BAM, SLUPP322)
- MT-2 Spray
- MT-1 Nasal Spray
- AOD-9604 Spray
- Phenibut (60 capsules)
- NAD+ 500MG Spray
- BPC-157 Spray
- Mazdutide 10MG
- AICAR 50MG
- Lipo-C 10ML
- Survodutide 10MG
- Methylene Blue 20MG
- 5-Amino-1MQ Capsules 50mg 60 cap
- LC-526 Metabolic Complex
- SSJ-9 Amino Matrix
- MITOCHONDRIAL POWER STACK
- SHRED MATRIX X4
- ANABOLIC SIGNALING MATRIX X3
- L-Carnitine 600MG / 10ML
- Cerebrolysin 60MG
- Oxytocin 5MG
- SELANK
- 5-Amino-1MQ
- VIP
- L-Glutathione 1500MG
- Acetic Acid Solution
- HCG 10000iu
- BAC Water 30ML
- BAC Water 10ML
- GLOW 70MG
- IGF-1 LR3 1MG
- Thymalin
- HCG (5000iu)
- Wolverine Stack Peptides – BPC-157 (5mg) / TB-500 (5mg)
- BAC Water 3ML
- Melanotan II (MT-2) 10MG
- SLU-PP-332 (5MG)
- KPV 10MG
- GHK-Cu
- KLOW 80 – GHK-Cu (50mg) / KPV (10mg) / BPC-157 (10mg) / TB500 (10mg)
- Gonadorelin
- L-Glutathione (200MG)
- SEMAX (10MG)
- Cagrilintide (10MG)
- DSIP – Delta Sleep‑Inducing Peptide (5MG)
- RETA GLP-3
- MK-ULTRA (MK-777)
- Kisspeptin-10
- VIP 10MG Spray
- Ipamorelin / CJC-1295 No Dac 10mg
- Tirz GLP-2
- L-Glutathione (500MG)
- GHK-Cu Spray
- P21 Peptide Spray
- N-Acetyl Selank Spray
- N-acetyl Semax Spray
- Adamax Nootropic – Nasal Spray
- Adalank Nootropic Peptide – Nasal Spray
- Pancragen 20 MG
- Prostamax 20 MG
- Livagen 20 MG
- Testagen 20 MG
- CardioCytogen 20 MG
- TB-4
- ARA‑290 10mg
- FOXO4-DRI (D-Retro-Inverso) 10mg
- KPV 250 MCG (60 Capsules)
- SLU-PP-332 (60 capsules, 250MCG)
- SS-31
- Tesamorelin (10MG)
- MK-677 12.5MG (60 capsules)
- AOD-9604
- IPAMORELIN 10MG
- Healing and Repair Research Blend (60 capsules)
- BPC-157 250MCG (60 capsules)
- Tesofensine 500MCG (100 capsules)
- PT-141 10MG
- NAD+
- TB-500 10MG
- SEMA GLP-1
- MOTS-c 10MG
- CJC-1295 (10MG)
- BPC-157 (10MG)
Capsules
- Dihexa
- Reduce (BAM, SLUPP322)
- Phenibut (60 capsules)
- Methylene Blue 20MG
- 5-Amino-1MQ Capsules 50mg 60 cap
- MK-ULTRA (MK-777)
- KPV 250 MCG (60 Capsules)
- SLU-PP-332 (60 capsules, 250MCG)
- MK-677 12.5MG (60 capsules)
- Healing and Repair Research Blend (60 capsules)
- BPC-157 250MCG (60 capsules)
- Tesofensine 500MCG (100 capsules)










